 Which can be reduced to the classic Wallis formula. Specifically, the calculation of Friedmann and Hagen resulted in an expression involving special mathematical functions called gamma functions leading to the formula And they were surprised to see that the ratio yielded-effectively-the Wallis formula for π. This enabled them to determine how the ratio of the Bohr values to the values obtained with the ‘tweaked’ variational principle changed as higher and higher energy levels were taken into account. They could then calculate the values for the different energy states and compare them with the values obtained by Bohr almost a century ago. However, Friedmann and Hagen were able to get around that by separating the problem into a series of l problems, each of which focused on the lowest energy level for a given orbital angular momentum quantum number, l. This is because the variational principle cannot ordinarily be applied if there are lower energy levels. Hagen got Friedmann involved to take advantage of her ability to work in both physics and mathematics.Īlthough applying the variational principle to calculate the ground state of a hydrogen atom is a relatively straightforward problem, its applicability to an excited state is far from obvious. Hagen also started thinking about whether it would be possible to apply this method to states other than the ground state. 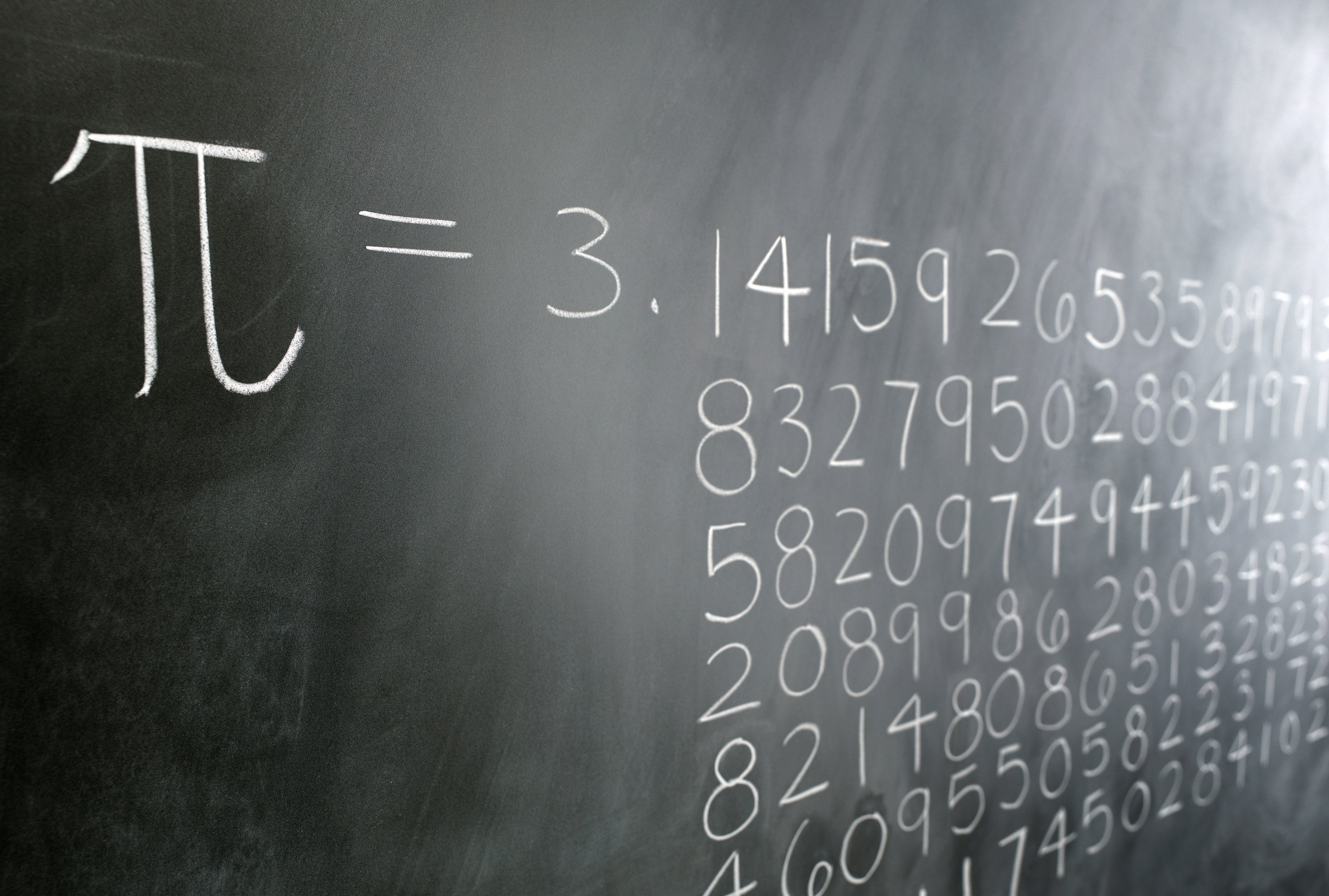
Like the Wallis formula, the variational principle dates back to the seventeenth century, one of its first appearances being the Principle of Least Time of mathematician Pierre de Fermat, a contemporary of Wallis. While the quantum calculations developed by Danish physicist Niels Bohr in the early twentieth century give accurate values for the energy states of hydrogen, Hagen wanted his students to use an alternate method-called the variational principle-to approximate the value for the ground state of the hydrogen atom. The discovery began in a quantum mechanics course taught by Carl Hagen, a professor of physics at the University of Rochester and one of the six physicists who predicted the existence of the Higgs boson. “The value of pi has taken on a mythical status, in part, because it’s impossible to write it down with 100 percent accuracy,” said Friedmann, “It cannot even be accurately expressed as a ratio of integers, and is, instead, best represented as a formula.”įriedmann did not set out to look for π nor for the Wallis formula. 
For Friedmann, discovering the Wallis formula for π in a quantum mechanics formula for the hydrogen atom’s energy states underscores π’s omnipresence in math and science. The Wallis formula-developed by British mathematician John Wallis in his book Arithmetica Infinitorum-defines π as the product of an infinite string of ratios made up of integers. “We found the classic seventeenth century Wallis formula for pi, making us the first to derive it from physics, in general, and quantum mechanics, in particular.” “We didn’t just find pi,” said Tamar Friedmann, a visiting assistant professor of mathematics and a research associate of high energy physics, and co-author of a paper published this week in the Journal of Mathematical Physics. 
But two University scientists were still surprised to find it lurking in a quantum mechanics formula for the energy states of the hydrogen atom. While most people associate the mathematical constant π (pi) with arcs and circles, mathematicians are accustomed to seeing it in a variety of fields. 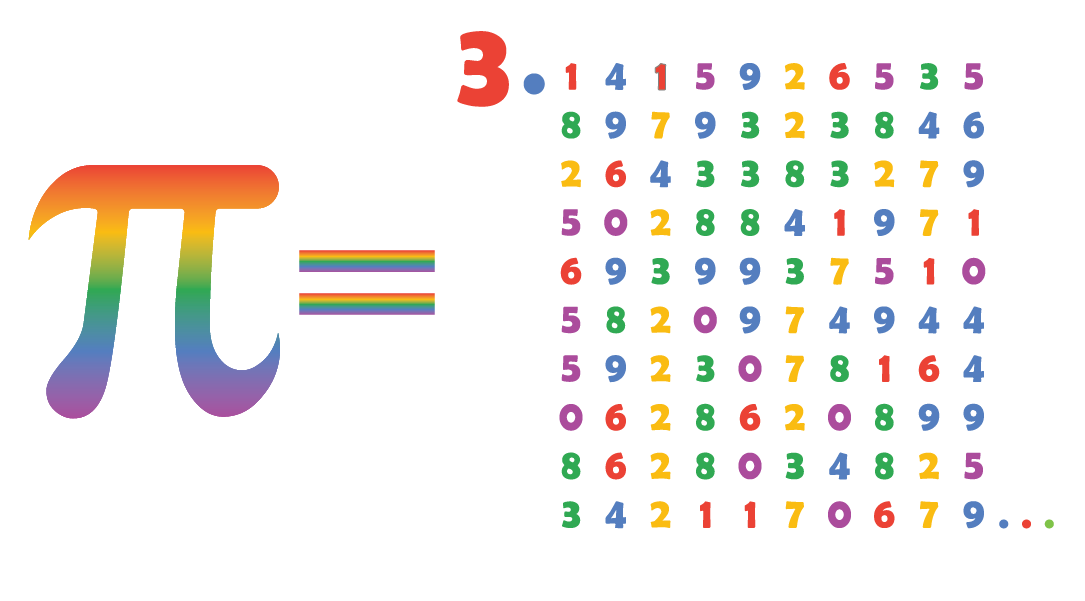
Wallis formula buried in quantum mechanics calculation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
